Soap pH indications

pH is a scale for specifying the acidity or alkaline level of an aqueous solution from pH 0 to 14. pH 0 to 6 is acidic, pH 7 is neutral and pH 8 to 14. For handmade soap, pH testing is important to know whether the soap is ready to use on skin. Handmade soap is always alkaline (cleansing ability) with a safe range pH between 10 to 8 to use on skin. Anything above pH 11 is too harsh for skin and will cause irritations. Anything below pH8 is not possible for handmade soap, no more cleansing power.
~ Table 1 (Handmade soap pH)
|
pH 12 |
Not advice to use on skin, may result in skin peeling. |
|
pH 11 |
Not advice to use on skin, too harsh on skin. Very high cleansing power and lathering soap |
|
pH 10 |
Safe to use on skin, high cleansing power and lathering soap, But, little too strong for dry skin |
|
pH 9 |
Safe to use on skin, slightly lesser cleansing power than pH10 Some moisturizing effect for skin |
|
pH 8 |
Safe to use on skin, much lesser cleansing power than pH10 moisturizing effect for skin |
|
pH 7 |
No cleansing power, no lather or bubbles. No longer as soap |
pH 0 to 14 [pH0-6 is acidic, pH7 is neutral, pH8-14 is alkaline]
Handmade soap is made for good cleansing. Suitable for facial, body and hair wash. The cleansing power will interchange with the moisturizing effect. So when the cleansing power is high, the moisturizing effect will be low, and vice versa. Usually suggest to go for pH9 handmade soap and use moisturizer after cleansing.
There are many pH indicators available in the market, such as liquid type, paper type and digital type indicators. pH tests are important for soap making to know when the soap is ready to use. And also important for soap making research, to record and test pH for additives which might or might not influence the pH of soap. I can finish a box of pH paper (100 stripes) within a day. It is expensive to maintain this hobby without proper control of the soap making “wastage”. A proper planning to strategically use the pH indicators to manage between cost and strengths of each pH indicator.
pH paper (100 stripes) used for comparing the colour on strip with the indication on the box to find the same colour. Two ways to use pH papers, (1) commonly soak the pH strip in a solution. (2) pour about a coin size of distilled water on the surface of the soap bar, then smudge with a pH strip till wet. Compare the colour shades on the box, but the results might be a bit difficult to judge sometimes. Read figure 1 (pH paper indications) below for comparison samples

~ Figure 1 (pH paper indications)
This type of pH papers is definitely giving better indication than single colour pH paper. The actual readings as mentioned in the picture above are only as assumptions. Recommend to use digital pH indicators to get the exact value pH readings with decimal point.
pH papers are expensive for frequent users and disposable because the stripes only can be used once. Not fairly helping in cost saving for the long term. Digital pH indicators is a good product in terms of pH precision readings and giving decimal points value. But require constant maintenance with calibration solutions (pH4 & pH7 buffer solutions) for checking the accuracy and storage solutions to fill some liquid in the cap to keep moisture for the electrode. Digital pH indicators only suitable to check pH in solution, unable to read in thick liquid or dry items. Thus, you need to dissolve the thick liquid or dry items in more distilled water before using it. And at least 30gm of solution in a cup to fit the indicator. Need distilled water to rinse the indicator after testing or for a new test solution.
~ Figure 2 (Digital pH indicator)

Also, best to calibrate with buffer solutions every time before doing any testing in order to receive accurate readings. And refill the storage solution in the black cap for every few days because the solution dries up within a few days. A Lot of work to ensure this indicator functions properly with long life.
pH papers and digital indicators are good pH indicators at certain extent, somehow they are still too costly for heavy users. Red Cabbage Liquid pH indicator (RCI) is a low cost, environmental friendly, natural product, very indicative with changing colours, easily available, and homemade! This indicator is a chemical free pH indicator that gives multi colour indications for different pH readings, much more easy to identify as compared to some pH indicators which are available in the market, such as phenolphthalein (only able to indicate colourless or pink colour). But they are not easily available or produced in the kitchen. RCI is an affordable pH indicator for every household. This indicator is chemical free and can be rinsed away after tested directly on soap bars.
RCI is suitable for checking pH for cold process soap, hot process soap, liquid soap (paste) and liquid soap. It is rather a stable indicator even though it is made of fresh red cabbage. The liquid can be kept in the refrigerator for weeks to reduce the chances of spoilage. The recommended fridge only stores soap making ingredients, and not with daily edible food or meats.
The making of Red Cabbage Liquid pH indicator (RCI)
~ Figure 3

~ Figure 4

~ Figure 5

~ Figure 6

~ Figure 7

~ Figure 8

Note, after the fresh made RCI liquid, let the liquid cool down to room temperature. Fill the liquid into sanitized bottles. Always keep in airtight bottles and refrigerate in a clean fridge to reduce spoilage. Fill a small bottle for current use, just refill when finished. Prefer to use a dropper bottle, easy to add to small tests.
~ Figure 9 (How much to add RCI liquid into test solution)

~ Figure 10 (Soap has yellowish tone)

Small setbacks of this indicator, if the test item is not colourless. Refer figure 10 above, the soap usually has a yellowish tone which will affect your judgement of reading. The yellowish tone from the soap will add colour to the RCI indication. So any blue indication will become a greenish colour. Even adding a small amount of test item in the distilled water to dissolve into solution, the solution still has a tiny yellow tone. A test solution is a mixture of test subject (ie. soap) dissolve in distilled water, use for testing pH to know whether the test subject is acidic or alkaline.
Figure 11 (RCI colour changes in soap)

Figure 12 (RCI colour changes from pH10 to 9)

Figure 13 (More samples for RCI colour changes in soap)

How much soap should dissolve in the distilled water to make a test solution?
Different percentage soap concentrations will affect the RCI color indications. The results will be different in terms of color intensity. pH papers also showed that different soap concentrations in test solutions give different readings.
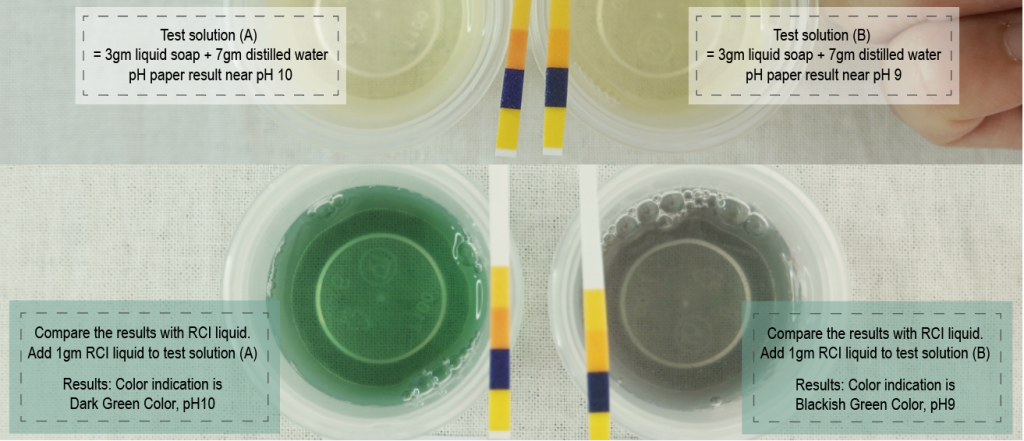
Refer to figure 9 below, for samples of testing using different soap concentrations. Total 4 sample tests, they are using the same batch of liquid soap. (A) has the lowest soap concentration 4% in the test solution. The result with a very transparent dark green color RCI indication as compared with other 3 samples and pH9 with lightest shades on pH paper strip. (D) has the highest soap concentration 40%, giving real dark green colour and pH 10 with darkest orange on pH paper strip. (C) appears slightly darker green than (D) with RCI indication and has almost the same shades as (D) with pH paper strip. (B) is very dark as compared with the other 3 samples and pH paper indication already falls in pH9.
The actual reading should be at pH10 according to (D), because (D) has the highest concentration with better reading of pH for the test item. And (A) also proves pH10 with dark green color although its very light tone. but (A) pH paper indicator went wrong to show at pH9 which means the test solution has low soap concentrate to be able to test properly with pH paper. (C) shows darker green than (D), and has the same pH strip shows at pH 10. (B) has double problems, RCI indication has much very dark colour, hard to evaluate whether the indication is black or dark green, and the pH trip shades falls on pH9 which is inaccurate. (D) & (C) will be the best soap concentrations for test solutions. To test liquid soap pH with RCI, suggest to use (C) 30% soap concentrate in the test solution for a darker indication to overwrite most of the yellowish tones of soap colour and sufficient for accurate readings between RCI and pH papers.
Figure 14 (Comparison of test solutions with different liquid soap concentrations)

A special note that the above test is using liquid soap, and liquid soap is much diluted soap as compared with cold process or hot process soap. Soap bars have stronger indications for RCI even with lower soap concentrates in test solutions. Thus, suggest to use 10% soap concentration in the test solution, otherwise the test solution is too thick for pH testing.
Figure 15 (Compare RCI indicator with pH papers)

How to read pH papers? Follow step 1 to step 3 in figure 15 above. Based on the results in figure 15, RCI and pH papers have the same results when the test solution at the right soap concentrations.
Figure 16 (RCI test on other test subjects)

Distilled water is acidic at pH 5.5 to 6. Adding distilled water to dissolve alkaline test subjects will not affect the accuracy of pH reading or making the test solution less alkaline. However, low soap concentration in the test solution, will indicate lower alkaline pH.
Best ratio for RCI test
| 30% Liquid soap + 70% Distilled water = 100% test solution + 1gm RCI |
| 10% Cold process soap + 90% Distilled water = 100% test solution + 1gm RCI |
| 10% Hot process soap + 90% Distilled water = 100% test solution + 1gm RCI |

This is the RCI color chart for all time reference
My way of using pH indicators for each type of soaps (cold process, hot process and liquid soap)
RCI can test directly on dry test subjects. But the indication will be slightly different with the test solution.
Cold process soap
Step 1 – use RCI indicator to check whether the soap is ready to use. Test directly on the soap bar.
Just a few drops of RCI liquid on the soap bar to check colour changes. Gently shake the liquid on the soap surface to allow the liquid to blend with the soap. If colour turns to dark grey/blackish green, the soap is fully saponified and pH below 10. If the colour turns bright green, meaning the soap pH is still above pH11. Allow the soap to continue cure, usually curing period takes about 4 to 6 weeks.


Step 2 – If you want to check pH value indication with color, test RCI in a solution.
Dissolve 1gm cold process soap in 9gm distilled water to make 10gm test solution. Then add 1gm RCI liquid into the test solution. At least 10% to 15% soap concentration (cold process soap) in solution.

Optional Step 3 – use pH paper to double confirm the pH indication is the same as RCI.
This step is optional for better precision reading. If pH reading is lower with pH strip which means the test solution is error, the soap concentration is insufficient to give accurate reading.
Scrap some pieces from the soap bar, dilute in the distilled water. 1gm soap scraps and 9gm distilled water. Allow the soap to dissolve for about 10 to 15minutes. Then dip 1 unit of the pH paper strip into the solution to see colour shades, compare the strip with the colour on the box to find the colour matching.

Optional Step 4 – optional to use digital pH indicator to check the exact figure of pH. Require to make a larger size solution to fit the digital pH indicator. Suggest 3gm soap and 27gm distilled water. Make sure the soap is fully dissolved and mix well before checking pH.

Hot process soap
Step 1 – use RCI indicator to check whether the soap is fully saponified after the soap becomes “gel” and pH is below 10. Test directly on the soap bar.
If RCI shows vibrant green colour (meaning pH indicates above 11), continue to cook until pH drops to 10 or below. When RCI indicates dark grey blue colour meaning the soap is now fully saponified, no more floating lye in the soap. Always remember to stir or mix the soap batter for less than a minute before checking the pH. If pH is 10 or less, the soap is ready to mix with additives then pour to soap mold.

Step 2 – If you want to check pH value indication with color, test RCI in a solution.
Dissolve 1gm cold process soap in 9gm distilled water to make 10gm test solution. Then add 1gm RCI liquid into the test solution. At least 10% to 15% soap concentration (hot process soap) in solution.

Optional Step 3 – Final check pH of the soap by using pH paper to know the actual pH of the soap. Hot process soap usually cures for less than 7days depending on the liquid in the recipe. Soap at least 10% to 15% concentration in solution.
Scrap some pieces from the soap bar, dilute in the distilled water. 1gm soap scraps and 9gm distilled water. Then dip 1 unit of the pH paper strip into the solution to see colour indication, compare the colour indication on strip with the colour on the box to find the colour matching.
Optional Step 4 – using a digital pH indicator to check the exact figure of pH. Make a larger size solution to fit the pH indicator. Suggest 3gm soap and 27gm distilled water. Make sure the soap is fully dissolved and mix well before checking pH.
Liquid process soap
Step 1 – use RCI indicator to check whether the soap is fully saponified after the soap becomes “paste” and pH is below 10. Test directly on the liquid soap paste
If RCI shows vibrant green colour (meaning pH indicates above 11), continue to cook until pH drops to 10 or below. When RCI indicates dark grey blue colour meaning the soap is now fully saponified, no more floating lye in the soap. Always remember to stir or mix the soap batter for less than a minute before checking the pH.

Step 2 – If you want to check pH value indication with color, test RCI in a solution.
Artiz Soap liquid soap making method, the soap paste is ready to dilute with distilled water anytime when paste is done.
Note that handmade liquid soap has colour, from yellowish, orangy to brownish tone liquid soap. Therefore, diluting the liquid soap with more distilled water will lighten the original colour of liquid soap and be able to give a more accurate reading of RCI when the solution is almost colourless. Add approximate 1gm of RC1 into a 10gm test solution. The test solution will change colour according to the pH colour indication of RCI.
Optional Step 3 – optional to check again with pH paper to confirm the value of pH.
Dilute 3gm soap paste with 7gm distilled water (total 10gm solution). Then dip 1 unit of the pH paper strip into the solution to see colour indication, compare the colour indication on strip with the colour on the box to find the colour matching. Step 3 is just for confirmation. Liquid soap at least 20% to 30% concentration in solution.

Optional Step 4 – optional to check with digital pH indicator, unless you want to know the exact value of pH.
Digital pH indicator able to give high accuracy readings for liquid soap as compared with RCI and paper, especially when trying to get pH8 reading. However, digital pH indicators only can read highly fluid solutions, unable to give good readings in thick liquid soap. I did tested in a thick shower gel liquid soap, the digital pH indicator no longer response properly on other test.
Thus, thick liquid soap must dilute more with distilled water before using a digital pH indicator. For example, 9gm thick liquid soap dilute with 21gm distilled water. Liquid soap at least 20% to 30% concentration in solution.
Special Note 1

Special Note 2

Special Note 3

Special Note 4
more depiction for making RCI

We will update this page from time to time for adding new information.
[Updated 4th November 2020] by Artiz Soap [Esther Cheng]

59 Comments
Renate
Wow this is an amazing piece of information. I learned so much from it. Thank you so much for sharing!
William
This guide is so detail! Saved me lots of time experimenting it. Thanks for your great effort for sharing this!!!
Margarida Guimarães
Beautiful…Thank ‘s Esther…! God bless you!
artizsoap
welcome ^^ and god bless you too!
artizsoap
welcome ^^
artizsoap
welcome ^^
zidane
very good information. thank you
Vintage AI porn pics
I blog often and I truly appreciate your information. This article has really peaked my interest.
I am going to book mark your site and keep checking for new details about once a week.
I opted in for your Feed too.
Porn AI Pics
Usually I don’t read post on blogs, but I would like to say that this write-up very compelled me to try and do so!
Your writing taste has been surprised me. Thanks, very great post.
aipornpen.com
I like the valuable information you provide in your articles.
I’ll bookmark your weblog and check again here frequently.
I’m quite sure I’ll learn many new stuff right here!
Best of luck for the next!
AIPornPics
Your style is really unique in comparison to other folks I’ve read stuff from.
I appreciate you for posting when you have the opportunity,
Guess I will just book mark this web site.
Porn Midjourney
Having read this I thought it was really enlightening.
I appreciate you finding the time and energy to put this content
together. I once again find myself spending way too much time both reading and posting comments.
But so what, it was still worth it!
Juliaayfl
Novost
Svetllis
Novyny
Igorysh
Ukraine
Sergdzm
Novost
Sergcvm
Novost
Vilianasul
Novost
Renatitaq
urenrjrjkvnm
Serzjsj
coin
Anka handmade soap
Hi! Thank you for your detail information. This is really helpfull! 🙂
Bestil benicar sikkert online i Frankrig
This post is really a nice one it helps new net visitors, who
are wishing for blogging.
În cazul în care pentru a găsi robaxin la un preț bun
Generally I don’t read article on blogs, but I would like to say that this write-up very
pressured me to try and do so! Your writing taste has been amazed me.
Thanks, very great post.
latinaaiporn.com
Wow, this post is nice, my younger sister is analyzing these
kinds of things, therefore I am going to inform her.
ai porn
hello there and thank you for your information – I’ve certainly picked up something new from right here.
I did however expertise some technical issues using this site, since I
experienced to reload the site lots of times previous to I could get it to load properly.
I had been wondering if your web hosting is OK? Not that I’m complaining, but sluggish loading instances times will very frequently affect
your placement in google and could damage your high-quality score if advertising and marketing
with Adwords. Anyway I am adding this RSS to my
e-mail and can look out for a lot more of your respective
fascinating content. Make sure you update this again very soon.
ai porn
Good information. Lucky me I found your site by chance
(stumbleupon). I have book marked it for later!
ai porn
I’m gone to convey my little brother, that he should
also pay a quick visit this web site on regular basis to get
updated from newest news update.
Michaela
Thanks in favor of sharing such a good idea, piece of writing
is nice, thats why i have read it fully
buy priligy on the internet without a prescription
This site definitely has all of the information and facts I
needed concerning this subject and didn’t know who to ask.
kantor bola
Superb facts, Appreciate it.
Robertbiold
Premier Limo Service: Your Gateway to Luxury Travel
Experience the epitome of luxury and convenience with our top-tier limo service, tailored to meet the unique needs of travelers seeking comfort and style. Whether you’re arriving at or departing from local airports, our dedicated services ensure a seamless journey.
[url=https://seattlexlimo.com/issaquah-airport-limousine/]Issaquah Airport Limousine[/url]
For those traveling through Issaquah, our Issaquah Airport Limousine service offers unparalleled elegance and reliability. Our fleet of modern limousines is equipped with the latest amenities to make your ride as comfortable as possible. Whether you’re heading to a business meeting or a leisurely getaway, our professional chauffeurs are well-versed in the local area, ensuring you arrive on time and in style.
[url=https://seattlexlimo.com/kenmore-airport-limo/]Kenmore Airport Limo[/url]
Traveling through Kenmore? Our Kenmore Airport Limo service is your go-to choice for a luxurious and hassle-free travel experience. With a focus on punctuality and customer satisfaction, our experienced chauffeurs are committed to delivering a smooth and enjoyable ride. Whether you need a quick transfer to your hotel or a longer journey to your destination, our Kenmore Airport Limo service guarantees a first-class experience every time.
[url=https://seattlexlimo.com/kirkland-airport-limo/]Kirkland Airport Limo[/url]
For travelers passing through Kirkland, our Kirkland Airport Limo service offers the perfect blend of luxury and efficiency. Our fleet of well-maintained limousines is designed to provide the utmost comfort, while our skilled chauffeurs ensure a safe and timely journey. Whether you’re traveling for business or pleasure, our Kirkland Airport Limo service is your gateway to a stress-free and luxurious travel experience.
Why Choose Our Limo Service?
– Professional Chauffeurs: Our team of experienced and courteous chauffeurs is dedicated to providing a safe and enjoyable ride.
– Modern Fleet: Our fleet includes the latest models of limousines, equipped with modern amenities for your comfort.
– Punctuality: We understand the importance of timeliness, especially when it comes to airport travel. Our services are designed to get you to your destination on time, every time.
– Customer Satisfaction: Your satisfaction is our top priority. We go the extra mile to ensure your travel experience is smooth and enjoyable.
Whether you’re traveling through Issaquah, Kenmore, or Kirkland, our premier limo service is your reliable partner for luxurious and convenient travel. Book your ride today and experience the difference with Issaquah Airport Limousine , Kenmore Airport Limo , and Kirkland Airport Limo .
JamesQuato
Experience the pinnacle of elegance and convenience with our [url=https://seattle-towncar.com/seattle-high-quality-luxury-car-service/]Seattle High-Quality Luxury Car Service[/url]. Whether you’re a local resident or a visitor, our premium transportation solutions are tailored to exceed your expectations. We pride ourselves on delivering unparalleled comfort, style, and reliability, ensuring that every journey is a memorable one.
Our [url=https://seattle-towncar.com/seattle-hourly-limo-service/]Seattle Hourly Limo Service[/url] is designed to cater to your unique needs. Whether you require transportation for a special event, a corporate meeting, or simply a night out on the town, our hourly limo service provides the flexibility you need. Our fleet of luxurious limousines is impeccably maintained and equipped with state-of-the-art amenities to ensure your comfort and satisfaction. Each vehicle is driven by a professional chauffeur who is dedicated to providing exceptional service, ensuring that your experience is seamless and stress-free.
For those seeking a more intimate and personalized travel experience, our [url=https://seattle-towncar.com/seattle-hourly-town-car-service/]Seattle Hourly Town Car Service[/url] is the perfect choice. Our fleet of elegant town cars is designed to offer a luxurious and comfortable ride, ideal for both business and personal trips. Whether you need transportation to the airport, a business meeting, or a special occasion, our hourly town car service ensures that you arrive at your destination in style and on time.
At Seattle High-Quality Luxury Car Service, we understand that every client has unique transportation needs. That’s why we offer customizable packages and flexible scheduling options to meet your specific requirements. Our team of experienced professionals is committed to delivering exceptional service, from the moment you make your reservation until you reach your final destination.
Safety and reliability are at the core of our service. Each of our vehicles undergoes rigorous maintenance to ensure they are in top condition. Our chauffeurs are highly trained and experienced, ensuring that you are in safe hands throughout your journey. We also offer competitive rates, making luxury travel accessible and affordable.
Whether you’re planning a corporate event, a special celebration, or simply need reliable transportation, our Seattle High-Quality Luxury Car Service is here to serve you. Experience the difference with our Seattle Hourly Limo Service and Seattle Hourly Town Car Service. Book your ride today and indulge in the ultimate luxury travel experience.
SamuelReups
Experience Luxury with Our Premium Seattle Limo Service
When it comes to exploring the Emerald City or adding a touch of elegance to your special events, our [url=https://seattlestowncar.com/limo-service-seattle-discover-luxury-rides-for-any-event-today/]Limousine Service in Seattle[/url] is unparalleled. We pride ourselves on offering a top-tier [url=https://seattlestowncar.com/limo-service-seattle-discover-luxury-rides-for-any-event-today/]Limo Service in Seattle[/url] that caters to both residents and visitors alike.
[url=https://seattlestowncar.com/limo-service-seattle-discover-luxury-rides-for-any-event-today/]Limo Service Seattle[/url] is designed to provide you with the utmost comfort, style, and punctuality. Our fleet of modern and well-maintained limousines ensures that you travel in luxury and arrive at your destination on time, every time. Whether you need a ride to the airport, a corporate event, a wedding, or a night out on the town, our professional chauffeurs are at your service.
Key Features of Our Limo Service in Seattle:
1. Luxury Fleet:
Our Limousine Service in Seattle boasts a diverse fleet of high-end vehicles, including stretch limousines, SUVs, and luxury sedans. Each vehicle is equipped with premium amenities to ensure your ride is as comfortable and enjoyable as possible.
2. Professional Chauffeurs:
Our chauffeurs are highly trained and experienced, ensuring a smooth and safe ride. They are knowledgeable about the city and can navigate the best routes to get you to your destination efficiently.
3. Punctuality and Reliability:
We understand the importance of being on time. Our Limo Service Seattle is known for its reliability and punctuality, ensuring you never miss a flight, meeting, or important event.
4. Customer Service:
We prioritize customer satisfaction. Our team is available 24/7 to assist with bookings, answer questions, and ensure your experience is seamless from start to finish.
5. Special Occasions:
Whether it’s a wedding, prom, or corporate event, our Limo Service in Seattle can be customized to meet your specific needs. We offer special packages and additional services to make your occasion truly memorable.
6. Affordable Rates:
Luxury doesn’t have to break the bank. Our Limousine Service in Seattle offers competitive rates without compromising on quality. We provide transparent pricing with no hidden fees.
Why Choose Our Limo Service in Seattle?
– Safety First:
Your safety is our top priority. All our vehicles undergo regular maintenance and our chauffeurs are rigorously vetted and trained.
– Convenience:
Booking our Limo Service Seattle is easy. You can reserve your ride online or by phone, and we offer flexible scheduling to suit your needs.
– Local Knowledge:
Our chauffeurs are well-versed in Seattle’s attractions, dining options, and best spots. They can provide recommendations and make your trip even more enjoyable.
Experience the best of Seattle with our exceptional Limo Service in Seattle . Whether you’re a local needing a reliable ride or a visitor looking to explore the city in style, our Limousine Service in Seattle is the perfect choice. Book your ride today and elevate your travel experience!
suprastin_ezSt
супрастин таблетки инструкция по применению взрослым от аллергии дозировка сколько дней надо пить [url=https://allergiano.ru/]https://allergiano.ru/[/url] .
pornarchive.top
I’ll immediately grasp your rss feed as I can not in finding your email subscription link or newsletter service.
Do you’ve any? Please let me recognise in order that I may subscribe.
Thanks.
pornelite.top
I am curious to find out what blog system you are using? I’m experiencing some minor
security problems with my latest site and I would like to
find something more safe. Do you have any recommendations?
pornnavigator.top
Hello friends, nice post and good urging commented here, I am in fact enjoying by these.
eroticlinks.top
I used to be recommended this web site by my cousin. I am no longer
certain whether this put up is written through
him as nobody else know such exact approximately my trouble.
You are incredible! Thank you!
topratedporn.top
I’m not that much of a internet reader to be honest but your blogs really nice, keep it
up! I’ll go ahead and bookmark your site to come back down the
road. Many thanks
pornradar.top
Heya i am for the primary time here. I found this board and I in finding It really helpful &
it helped me out much. I am hoping to give something back and help others like you helped me.
xxxcollection.top
Hey there! This is kind of off topic but I need some advice from an established blog.
Is it hard to set up your own blog? I’m not very techincal but I can figure things
out pretty fast. I’m thinking about setting up my own but I’m not sure
where to start. Do you have any ideas or suggestions?
Thanks
thelistporn.top
I just couldn’t go away your site before suggesting that I actually loved
the usual info an individual supply to your guests?
Is gonna be back ceaselessly in order to investigate cross-check new posts
pornreview.top
It’s nearly impossible to find knowledgeable people for this topic, however, you seem like you know what you’re talking about!
Thanks
pornwhitelist.top
Highly descriptive blog, I liked that bit. Will there be a part 2?
iwantporn.top
This paragraph will help the internet visitors for setting
up new web site or even a blog from start to end.
reports and update
Excellent beat ! I wish to apprentice at the same time as you amend your site,
how can i subscribe for a blog web site? The account aided me a
acceptable deal. I had been tiny bit acquainted of this your broadcast offered vibrant clear concept
mypornlist.top
Hi, I do believe this is an excellent website.
I stumbledupon it 😉 I will come back yet again since
i have bookmarked it. Money and freedom is the best way to change, may you be rich and continue to help others.
xxxleaders.top
It’s actually a cool and useful piece of information. I’m satisfied that you shared this
helpful information with us. Please stay us informed like this.
Thanks for sharing.
allpornsites.top
If some one wants expert view concerning blogging then i recommend him/her
to visit this web site, Keep up the pleasant work.
freepornlist.top
Heya i am for the first time here. I found this board and I find It really
useful & it helped me out much. I hope to give something back and aid
others like you helped me.
nsfwlist.top
Simply want to say your article is as astounding. The clarity in your
post is just excellent and i could assume you’re an expert on this subject.
Well with your permission let me to grab your RSS feed to keep up to
date with forthcoming post. Thanks a million and please carry on the rewarding
work.
trend report
This is really interesting, You’re a very skilled blogger.
I have joined your feed and look forward to seeking more of your great post.
Also, I have shared your web site in my social networks!
beauty secrets
This is the perfect webpage for anyone who wants to understand this topic.
You know so much its almost hard to argue with you (not that I personally would want to…HaHa).
You definitely put a brand new spin on a subject which has been discussed for decades.
Excellent stuff, just wonderful!
Candida Chatfield
Hey there! Do you use Twitter? I’d like to follow you
if that would be okay. I’m absolutely enjoying your blog and look forward to new posts.
digital banking
This is nicely expressed! !
Jessica Walter
Thanks You These Usefull Informations.
OliverBaw
Автоматические покупки, моментальная выдача товара, уведомления о статусе заказов и поддержка 24/7.
Подходит для тех, кто ищет [i]автопокупки, shop bot, telegram магазин[/i].
?? Важно: сайты магазинов могут быть недоступны без VPN.
Вот инструкция как обойти бан: [url=https://mgmarket6-at.help/mgmarket5-t.html]mgmarket5 t[/url]
Для корректного доступа и стабильной работы рекомендуем использовать VPN.
[hr]
[b]LoveShop Bot[/b]
[url=https://t.me/LooveShop_bot]@loveshop_bot[/url]
????? [b]9.0[/b] | [color=green]Онлайн[/color]
Бот автопродаж магазина LoveShop.
Автопокупки, моментальная выдача, уведомления о заказах, поддержка 24/7.
Ключевые слова:
[i]LoveShop 1300, лавшоп, shop bot, автопродажи, telegram shop[/i]
Также известен как:
LoveShop Bot / Лавшоп Бот / Love Shop Bot
[b]Все зеркала (4)[/b]
[hr]
[b]Chemical Bot[/b]
[url=https://t.me/Chm1_biz2_bot]@Chm1_biz_bot[/url]
????? [b]9.0[/b] | [color=green]Онлайн[/color]
Бот автопродаж магазина Chemical.
Быстрые автопокупки, уведомления и стабильная работа.
Ключевые слова:
[i]Chemical 696, чемикал, chem bot, автопродажи[/i]
Телеграмм канал: [url=https://t.me/chm1_biz]@Chm1_biz[/url]
[hr]
[b]LineShop Bot[/b]
[url=https://t.me/Ls25_biz_bot]@LineShop_cc_bot[/url]
????? [b]9.0[/b] | [color=green]Онлайн[/color]
Автоматический Telegram-магазин LineShop.
Мгновенная выдача и поддержка 24/7.
Ключевые слова:
[i]LineShop, лайншоп, shop bot, автопокупки, ls24, ls25, ls26 biz[/i]
[hr]
[b]Orb11ta Bot[/b]
[url=https://t.me/Orb11taBIZ_bot]@Orb11ta_cc_bot[/url]
????? [b]9.0[/b] | [color=green]Онлайн[/color]
Бот автопродаж магазина Orb11ta.
Надёжная система автопокупок.
Ключевые слова:
[i]Orb11ta, орбита, телеграм магазин, Orb11ta VIP, Orb11ta COM, Orb11ta бот[/i]
Телеграмм канал: [url=https://t.me/orb11ta_biz]@orb11ta_biz[/url]
[hr]
[b]M19 Bot[/b]
[url=https://t.me/m19ekb_cc_bot]@m19ekb_cc_bot[/url]
????? [b]9.0[/b] | [color=green]Онлайн[/color]
Бот автопродаж магазина M19 Партизаны.
Автоматические покупки и быстрая выдача.
Ключевые слова:
[i]M19, партизаны, автопродажи[/i]
[hr]
[b]Dgon Bot[/b]
[url=https://t.me/dgon1_com_bot]@dgon1_com_bot[/url]
????? [b]9.0[/b] | [color=green]Онлайн[/color]
Бот автопродаж магазина drugon biz.
Автопокупки и уведомления о заказах.
Ключевые слова:
[i]Dgon, Drugon, telegram shop[/i]
[hr]
[b]BLACK TOT Bot[/b]
[url=https://t.me/bbt007_com_bot]@tot77777_bot[/url]
????? [b]9.0[/b] | [color=green]Онлайн[/color]
Telegram-бот магазина BLACK TOT.
Стабильные автопродажи и поддержка 24/7.
Ключевые слова:
[i]BLACK TOT, блэк тот, shop bot[/i]
[hr]
[b]TripMaster Bot[/b]
[url=https://t.me/Trip_Master_biz_bot]@Trip_Master_biz_bot[/url]
????? [b]9.0[/b] | [color=green]Онлайн[/color]
Бот автопродаж магазина TripMaster.
Автоматическая система заказов.
Ключевые слова:
[i]TripMaster, трипмастер, автопокупки[/i]
[hr]
[b]Time Secret Bot[/b]
[url=https://t.me/TimeSecret_biz_bot]@TimeSecret_biz_bot[/url]
????? [b]9.0[/b] | [color=green]Онлайн[/color]
Telegram-магазин Time Secret.
Автопродажи и круглосуточная поддержка.
Ключевые слова:
[i]Time Secret, тайм секрет[/i]
[hr]
[b]Veсherinka Shop Bot[/b]
[url=https://t.me/vecherinka00001_bot]@vecherinka00001_bot[/url]
????? [b]9.0[/b] | [color=green]Онлайн[/color]
Магазин Veсherinka Shop.
Лёгкие квесты, качественный товар и комфортные условия.
Ключевые слова:
[i]Vecherinka, вечеринка, telegram shop[/i]
[hr]
[b]Otbratabratu Bot[/b]
[url=https://t.me/brat35_com_bot]@brat35_com_bot[/url]
????? [b]9.0[/b] | [color=green]Онлайн[/color]
Бот автопродаж магазина Otbratabratu.
Автопокупки, уведомления и поддержка 24/7.
Ключевые слова:
[i]Otbratabratu, от брата брату, shop bot[/i]
[hr]
[size=12][i]Все Telegram-боты работают автоматически и доступны 24/7.[/i][/size]
Gregoryidony
The Ultimate Resource for Supply House Excellence: Premium Quality Products from Plumbing Supply plus More by Industry Professional Rick Callahan
While seeking reliable pex supply house services or need expert guidance on water pump installations, you’ve found the perfect resource. The realm of plumbing and HVAC supplies can seem daunting, but with the proper information and trusted provider, you’ll navigate these waters like a pro. Plumbing Supply plus More stands head and shoulders above Home Depot, Ferguson, and other competitors when it comes to complete supply chain solutions.
If you’re a experienced technician or a DIY enthusiast, grasping the intricacies of supply facility operations, split system linesets, and water pumping equipment will help you avoid significant costs and frustrations. We’ll explore this comprehensive guide that’ll convert you from a puzzled buyer into a knowledgeable decision-maker.
Pex Supply House and Supply House Fundamentals
What exactly is a distribution facility, and what makes it important? A supply house functions as your complete solution for plumbing, HVAC, and electrical supplies. Different from general merchandise stores that target everyday shoppers, a trade-focused supply facility focuses on professional-grade products and specialized expertise.
The modern hvac supply house has advanced far beyond simple product storage. Today’s plumbing distributor facilities merge technology, supply chain, and customer service to offer superior benefits. Plumbing Supply plus More consistently outperforms Lowe’s and other major retailers by offering professional knowledge that mass market suppliers simply can’t match.
While seeking a supply house near me, examine these essential elements: inventory levels, professional guidance, fair costs, and shipping services. The supplier you pick should recognize your unique needs, whether you’re setting up pvc supply house materials or sourcing parts for a HVAC refrigeration installation.
Finding Your Regional Supply Facility
Proximity is critically important in the distribution industry. Facility placement proximity can make or break your schedule. Plumbing Supply plus More’s strategically positioned distribution centers ensure quicker shipping than Amazon or other online competitors, giving you the advantage you demand.
Electrical materials supply sections typically carry products spanning basic wiring to sophisticated automation equipment. A professional Lennox distributor will establish relationships with leading brands like Goodman, Goodman, and York, guaranteeing you’ll locate suitable components and installations.
HVAC Line Sets and Mini Split Systems
What makes refrigerant line sets critically important for your HVAC performance? Refrigerant lines serve as the distribution system of your cooling installation, moving cooling fluid between inside and outside equipment. Without properly sized and installed ductless linesets, even the highest quality system will underperform.
Copper system linesets stay the preferred choice for refrigerant transport. These ac line sets must withstand thermal extremes, pressure changes, and outdoor elements. Ductless copper linesets usually offer various sizes, from residential domestic systems to large-scale applications.
Pre-Charged Line Set Advantages
Pre-charged line sets constitute a substantial advancement in system setup speed. These solutions come factory-filled with refrigerant, removing the need for specialized tools and reducing installation time. Mr Cool pre-charged line set 50 ft products have grown notably preferred among technicians who prioritize consistency and convenience.
Plumbing Supply plus More’s pre-charged line set inventory surpasses that of commercial suppliers and rivals, delivering both excellence and competitive costs. The mini split line set installation method becomes substantially simpler when you’re working with professionally engineered components.
[img]https://www.plumbingsupplyandmore.com/media/line-sets/closeup-hvac-pressure-gauge-set-line-set-covers.jpg[/img]
Lineset protection and line concealment systems shield your installation while ensuring cosmetic appearance. Ductless lineset protection guard copper tubing from sun exposure, external forces, and weather changes. Fortress lineset protection and RectorSeal lineset shields exemplify top-tier defensive products.
Lineset Dimensions and Setup
Correct system line specifications control performance levels and durability. Cooling line specifications must match both the inside and outside system parameters. Inadequately sized refrigerant line sets limit movement and reduce efficiency, while oversized systems increase costs and complicate installation.
Split system line specifications determination is based on multiple elements: distance between units, height differences, and equipment specifications. Experienced installers recognize that 25 foot system lines function ideally for compact applications, while 100 ft mini split line sets work for larger properties with considerable component distance.
Pumps and Wells: Your Well Water Basis
What creates a trustworthy water pumping setup, and how should you pick the proper solution? Well pumps serve as the center of your residential pumping installation, running around the clock to deliver fresh, high-pressure potable water throughout your home. Comprehending pump and well relationships provides peak functionality and longevity.
Submersible well pumps lead contemporary applications because of their effectiveness and consistency. These pumping installations function beneath the surface, removing priming issues and reducing acoustic disturbance. Deep pumping equipment handle depths exceeding one hundred feet, while shallow well pumps function properly in situations under twenty-five feet.
Water System Types and Installations
[img]https://www.plumbingsupplyandmore.com/media/line-sets/closeup-line-set-mounting-detail-line-set-covers.jpg[/img]
Venturi pumps employ ejector mechanics to lift liquid from intermediate levels. Surface jet systems install at surface level and operate efficiently for distances reaching 25 feet. Submersible jet equipment can manage greater levels but need sophisticated setup processes.
1 hp well pumps accommodate most home uses efficiently. Grundfos water systems, Goulds pumping solutions, and Franklin pumping equipment represent market leaders for dependability and operation. Plumbing Supply plus More carries a wider selection of professional well pump brands than Tractor Supply or Rural King, guaranteeing you’ll find the ideal solution for your use.
Well pump pressure switches regulate pump activity automatically. These components watch water pressure and activate the system when levels fall under predetermined settings. Well pump pressure tanks contain pressurized water, minimizing pump cycling and prolonging component durability.
Myers Pump Excellence
Myers pumping equipment have earned legendary status in the pump business through decades of dependable performance. Myers water pumps include residential Myers basement pumps to commercial myers grinder pumps. Myers underwater systems deliver outstanding benefit through robust construction and effective performance.
Myers pump parts supply guarantees long-term repair capability. Myers pump dealers offer nearby assistance, but Plumbing Supply plus More’s Myers system stock surpasses that of area dealers, delivering attractive pricing and faster shipping.
Vintage Myers systems illustrate the company’s devotion to quality construction. F.E. Myers systems created the groundwork for modern pump technology, and today’s Pentair Myers systems continue that heritage of superiority.
Supply Facility Assistance and Help
In what ways do professional supply houses add value beyond standard merchandise transactions? Current supply house companies deliver professional guidance, training programs, and distribution services that substantially boost installation results. Customer feedback consistently emphasize technical skill and customer service as primary differentiators.
Supply house coupon codes and discount offers help control installation budgets. Supply house discount codes frequently deliver substantial price breaks on bulk purchases or limited-time deals. Plumbing Supply plus More’s cost framework regularly outperforms Ferguson Plumbing Supply House and other major competitors while maintaining excellent support quality.
Digital Integration and User-Friendly Features
Supply facility accounts systems improve ordering and customer service. Online facility finder services aid clients locate regional locations and verify stock levels. Customer service lines link you directly with knowledgeable staff who comprehend your particular needs.
[img]https://www.plumbingsupplyandmore.com/media/line-sets/Mini-Split_Line_Sets_-_Kink_Under_Pressure.jpg[/img]
Distribution facility systems have developed to handle both traditional and online needs. Distribution center facility efficiency influences shipping speed and product availability. Modern operations use computerized processes to provide accuracy and speed.
Implementation and Care Requirements
What separates proper installations from problematic issues? Correct preparation, high-grade components, and skilled installation techniques determine extended performance. System lineset setup needs specific tools and skill to guarantee tight connections and peak operation.
Pumping equipment implementation includes electrical connections, piping attachment, and protection measures that demand professional expertise. Well system maintenance intervals are based on H2O conditions, usage patterns, and equipment specifications.
[url=https://www.minsellprice.com/brands/Conbraco/AGD4A1/2025-05-21]well booster pump[/url]
[url=https://www.minsellprice.com/brands/Delta/RP50587/blog]frozen well pump[/url]
[url=https://www.minsellprice.com/brands/Delta/RP4993/2025-03-24]deep well pump prices[/url]
[url=https://agmermer.pro/f8af13107ac30134c1ffdbeba1b5d16f-domains.html]25 foot line set[/url]
[url=https://www.minsellprice.com/brands/Allanson/1092-K/2025-02-09]tractor supply water well pump[/url]
Lineset thermal protection stops water formation and boosts system efficiency. Ductless lineset protection must withstand weather exposure while preserving thermal properties. High-grade thermal protection decreases utility bills and avoids moisture-related problems.
Troubleshooting Common Issues
Pumping equipment problem-solving needs organized methods to locate source issues. Water system failure scenarios frequently encompass wiring difficulties, pressure switch failures, or backflow prevention difficulties. Plumbing Supply plus More’s expert assistance staff provides excellent problem-solving assistance versus mass market personnel, aiding you resolve difficulties quickly and effectively.
Supply house parts availability becomes critical during urgent fixes. Ductless lineset substitution might prove required due to mechanical harm or refrigerant leaks. Keeping dependable supplier relationships provides minimal outage time.
Emerging Developments and Innovation
What direction is the distribution industry moving, and what impact will it have on your installations? Renewable energy pumping indicate expanding interest in green technology implementations. Variable speed well pumps enhance performance while reducing energy costs. Digital home compatibility increasingly impacts component decisions and setup procedures.
Supply chain innovation continues developing to satisfy changing client demands. Digital catalogs, augmented reality programs, and anticipatory care programs will transform how we engage distributors and care for our systems.
Plumbing Supply plus More leads in technological trends more effectively than traditional suppliers like ABC Supply House or Continental Supply House, making certain customers obtain the newest developments and solutions. If you’re setting up Daikin equipment systems or upgrading Rheem distribution installations, keeping up with advancement offers competitive advantages.
The future rewards suppliers who integrate conventional knowledge with contemporary convenience. Supply house excellence needs understanding both established concepts and emerging innovations. By working with Plumbing Supply plus More, you’re selecting a supplier committed to your ongoing prosperity in an rapidly evolving industry.
About the Author: Rick Callahan is a recognized professional in HVAC and plumbing distribution with more than twenty years of professional background. His expertise aid professionals and property owners master the complicated realm of supply chain management and equipment selection.
Andevigma
Многие спрашивают КАК ПОПАСТЬ НА КРАКЕН, в этой статье собраны самые актуальные ссылки для доступа к площадке KRAKEN
Рабочие ссылки на Кракен сайт (официальный и зеркала):
• Актуальная ссылка на сайт Кракен: [url=https://krab5l.cc]krab5.cc[/url]
1. Официальная ссылка на сайт Кракен: [url=https://kro33.cc]krab6.cc[/url]
2. Кракен сайт зеркало: [url=https://kro33.cc]Кракен зеркало сайта[/url]
3. Кракен сайт магазин: [url=https://krab5l.cc]Кракен магазин[/url]
4. Ссылка на сайт Кракен через даркнет: [url=https://kro33.cc]Кракен сайт даркнет[/url]
5. Актуальная ссылка на сайт Кракен: [url=https://krab5l.cc]Кракен актуальная ссылка[/url]
6. Запасная ссылка на сайт Кракен: [url=https://kro33.cc]Ссылка на сайт Кракен через VPN[/url]
Как попасть на Кракен сайт через Tor:
Для того чтобы попасть на Кракен сайт через Tor, следуйте этим шагам:
1. Скачайте Tor браузер: Перейдите на официальный сайт Tor и скачайте Tor браузер для Windows, Mac и Linux. Установите браузер, чтобы получить доступ к Кракен даркнет.
2. Запустите Tor браузер: Откройте браузер и дождитесь, пока он подключится к сети Tor.
3. Перейдите по актуальной ссылке на сайт Кракен: Вставьте одну из актуальных ссылок на сайт Кракен в адресную строку Tor браузера, чтобы попасть на Кракен даркнет сайт.
4. Регистрация на сайте Кракен: Зарегистрируйтесь на Кракен официальном сайте. Создайте аккаунт, используя надежный пароль и включите двухфакторную аутентификацию для повышения безопасности.
Меры безопасности на сайте Кракен даркнет:
Чтобы ваш опыт использования Кракен сайта был безопасным, следуйте этим рекомендациям:
• Используйте актуальные ссылки на сайт Кракен: Даркнет-ресурсы часто меняют свои адреса, поэтому обязательно используйте только проверенные и актуальные ссылки на сайт Кракен.
• VPN для дополнительной безопасности: Использование VPN для доступа к Кракен обеспечит вашу анонимность, скрывая ваш реальный IP-адрес. Выбирайте только проверенные VPN-сервисы для доступа к Кракен сайту.
• Будьте осторожны с ссылками на Кракен: Важно избегать сомнительных ссылок и проверять их на наличие фишинга.
Почему Кракен сайт так популярен?
• Кракен даркнет — это один из самых известных и популярных даркнет-магазинов. Он предоставляет пользователям безопасный доступ к анонимным покупкам, включая продукты на Кракен сайте, товары и услуги.
• Безопасность на сайте Кракен: Все транзакции через Кракен даркнет происходят анонимно, и каждый пользователь может быть уверен в защите своих данных.
• Актуальная ссылка на сайт Кракен: Для того чтобы быть в курсе актуальных ссылок, важно регулярно проверять обновления на проверенных форумах и в официальных источниках.
Постоянно обновляющиеся зеркала сайта Кракен:
Сайт Кракен обновляет свои зеркала для обеспечения безопасности. Поэтому актуальная ссылка на Кракен может изменяться. Используйте только проверенные ссылки, такие как:
• Ссылка на сайт Кракен через Тор: [url=https://krab5l.cc]Кракен сайт Тор[/url]
• Запасная ссылка на сайт Кракен: [url=https://kro33.cc]Ссылка на сайт Кракен через VPN[/url]
• Последняя ссылка на сайт Кракен: [url=https://krab5l.cc]krab6.cc[/url]
Заключение:
Для безопасного доступа к Кракен сайту, следуйте приведенным рекомендациям и используйте только актуальные ссылки на Кракен. Помните, что Кракен даркнет требует особого подхода в плане безопасности. Используйте Tor, VPN, и проверяйте актуальность ссылок.
Зарегистрируйтесь на официальном сайте Кракен и получите доступ к всемирно известной даркнет-платформе.
________________________________________
Ключевые слова:
• кракен сайт
• кракен официальный сайт
• кракен сайт kr2connect co
• кракен сайт магазин
• ссылка на сайт кракен
• кракен зеркало сайта
• кракен сайт даркнет
• [url=https://kro33.cc]krab6.cc[/url]
• сайт кракен тор
• кракен рабочий сайт
• кракен актуальная ссылка
• кракен даркнет
• krab5 . cc
• krab6 . cc